For most of the past two decades, Indian men diagnosed with advanced prostate cancer had a fairly narrow set of treatment options — hormone therapy, chemotherapy if the cancer progressed, and more recently oral targeted medicines like Enzalutamide (Glenza) and Abiraterone (Abirapro). These are genuinely effective medicines, but they work for a limited time in many patients. Once castration resistance develops and the cancer starts growing despite hormone suppression, the options narrow quickly.
That is changing in 2026, driven by one of the most clinically significant advances in prostate cancer treatment in recent memory: radioligand therapy using Lutetium-177 PSMA — a precision treatment that seeks out and destroys prostate cancer cells throughout the body using targeted radiation, while leaving surrounding healthy tissue largely unaffected.
This article explains what Lu-177 PSMA therapy is, what the latest 2026 clinical data shows, who qualifies for it, and what Indian oncologists and patients need to understand about accessing it.
What Is Lutetium-177 PSMA Therapy?
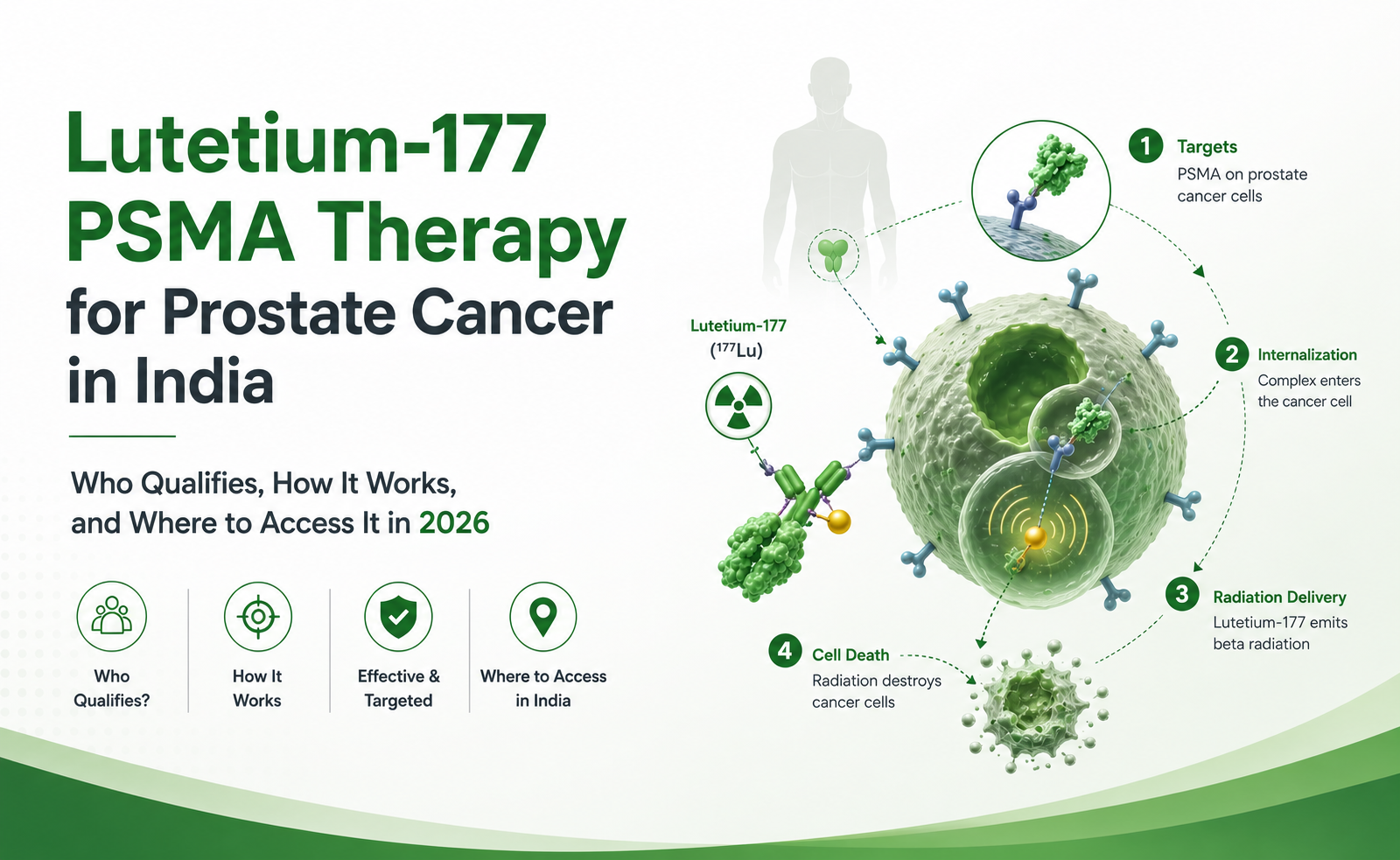
Lutetium-177 PSMA therapy is a radioligand therapy (RLT) — a class of treatments that combines a targeting molecule with a radioactive payload. The targeting molecule (vipivotide tetraxetan, also known as PSMA-617) binds with very high affinity to PSMA — prostate-specific membrane antigen — a protein expressed at 100 to 1,000 times higher levels on prostate cancer cells than on normal tissue. Once bound, the attached Lutetium-177 isotope emits beta particles that deliver lethal radiation directly to the cancer cell from within.
What makes this approach clinically powerful is its systemic reach. A single intravenous infusion distributes throughout the body via the bloodstream, binding to every PSMA-positive metastatic deposit simultaneously — bone metastases, lymph node deposits, soft tissue disease — treating disseminated cancer in a way that surgery or conventional radiotherapy simply cannot.
The treatment has been commercially available as Pivikto (Lutetium Lu-177 Vipivotide Tetraxetan) from Novartis, and requires administration in a licensed nuclear medicine facility under AERB-compliant radiation safety protocols.
What the Latest 2026 Data Shows
Two significant clinical developments in early 2026 have substantially strengthened the case for Lu-177 PSMA therapy.
Real-World Data Confirms Clinical Trial Outcomes — ASCO GU, February 2026
At the ASCO Genitourinary Cancers Symposium held in San Francisco on February 26, 2026, Novartis presented new real-world data from the PRECISION platform — a database integrating data from over 56,500 patients with metastatic prostate cancer. In taxane-naive metastatic castration-resistant prostate cancer (mCRPC) patients who had received prior ARPI therapy, real-world median progression-free survival with Lu-177 PSMA therapy was 13.5 months — closely consistent with the PSMAfore trial results that supported the FDA’s expanded approval.
This real-world consistency matters enormously for Indian oncologists. Clinical trial populations are carefully selected and monitored in ways that routine practice cannot always replicate. When real-world outcomes match trial outcomes in a large dataset, it gives confidence that the treatment will perform similarly in diverse, everyday clinical settings — including Indian oncology centres.
PSMAddition Trial — Moving Into Earlier Disease, 2026
The Phase 3 PSMAddition trial tested adding Lu-177 PSMA therapy to standard hormone therapy in patients with metastatic hormone-sensitive prostate cancer (mHSPC) — an earlier disease stage than currently approved. Results showed a statistically significant and clinically meaningful improvement in radiographic progression-free survival (rPFS) vs hormone therapy alone, with a positive trend in overall survival. Novartis has confirmed plans to submit for regulatory approval in this earlier indication in the second half of 2026.
This is a genuine paradigm shift. Currently, Lu-177 PSMA therapy is approved for castration-resistant patients who have already received an ARPI. The PSMAddition data suggests it may soon be appropriate much earlier — before castration resistance develops — potentially expanding eligibility to significantly more Indian patients.
How It Fits Into the Prostate Cancer Treatment Pathway
For Indian oncologists managing advanced prostate cancer, the current and emerging treatment sequence looks like this:
Stage 1 — Metastatic Hormone-Sensitive (mHSPC):
Androgen deprivation therapy (ADT) — Zoladex (Goserelin) or Firmagon (Degarelix) — combined with an ARPI: Glenza (Enzalutamide) or Abirapro (Abiraterone). PSMAddition data may add Lu-177 PSMA here in 2026.
Stage 2 — After ARPI, Before Chemotherapy (PSMAfore indication — currently approved):
For PSMA-positive mCRPC patients appropriate to delay taxane chemotherapy — Lu-177 PSMA therapy as an alternative to a second ARPI switch, based on the PSMAfore trial.
Stage 3 — After ARPI and Taxane Chemotherapy (original VISION indication):
For PSMA-positive mCRPC patients who have received both prior ARPI and prior taxane-based chemotherapy — the setting of the landmark VISION trial which demonstrated 38% reduction in mortality risk vs best supportive care.
For most Indian patients currently entering the treatment pathway, oncologists should be considering PSMA PET/CT imaging much earlier than previously necessary — both to select patients appropriately and to ensure imaging capability is in place when Lu-177 PSMA therapy becomes the next treatment step.
Who Qualifies for Lu-177 PSMA Therapy in 2026?
Eligibility is based on the following criteria:
1. Confirmed PSMA-Positive Disease by PSMA PET/CT
PSMA PET/CT imaging using Gallium-68 PSMA or Fluorine-18 PSMA tracer is mandatory before treatment. At least one PSMA-positive lesion with uptake greater than liver on PET/CT is required. Patients with PSMA-negative or very low PSMA-expressing disease do not respond and should not receive this treatment.
2. Metastatic Castration-Resistant Prostate Cancer (mCRPC)
Testosterone suppressed to castrate levels with ongoing ADT, with documented radiographic or PSA progression confirming castration resistance.
3. Prior ARPI Therapy
Prior treatment with an androgen receptor pathway inhibitor — Glenza (Enzalutamide) or Abirapro (Abiraterone Acetate) — is required under the current PSMAfore-based approval. The original VISION indication additionally required prior taxane chemotherapy.
4. Adequate Performance Status and Organ Function
ECOG performance status 0–2, ANC ≥1.5 × 10⁹/L, platelets ≥100 × 10⁹/L, haemoglobin ≥9 g/dL, and creatinine clearance ≥30 mL/min.
5. Access to a Licensed Nuclear Medicine Facility
Lu-177 PSMA therapy must be administered by trained nuclear medicine physicians and radiopharmacists in an AERB-licensed facility. It cannot be given in a standard oncology ward or infusion unit. SPECT/CT capability for post-dose imaging is also required.
The Treatment Regimen
- Dose: 7.4 GBq (200 mCi) intravenous infusion over approximately 30 minutes
- Schedule: Every 6 weeks for up to 6 cycles
- Companion ADT: Zoladex (Goserelin) or Firmagon (Degarelix) must be continued throughout all 6 cycles
- Monitoring: CBC, renal function, and PSA before each cycle; imaging assessment every 2 cycles
- Post-dose precautions: Radiation safety protocols for approximately 2 weeks after each infusion — patients must maintain distance from pregnant women and young children
Side Effects — What Patients Should Expect
Lu-177 PSMA therapy has a characterised and generally manageable side effect profile. The most common include:
- Dry mouth (xerostomia) — approximately 39% — the most distinctive side effect, occurring because salivary glands express low levels of PSMA and receive minor radiation exposure. Generally Grade 1–2.
- Fatigue — 43%
- Nausea — 35%
- Anaemia and haematological effects — 32% anaemia — from bone marrow radiation when extensive bone metastases are present. Mandatory CBC monitoring before each cycle.
- Renal toxicity — Kidneys are the primary elimination pathway. Monitor creatinine clearance before each cycle. Contraindicated if clearance <30 mL/min.
In the TheraP trial, which directly compared Lu-177 PSMA therapy against Cabazitaxel chemotherapy in post-Docetaxel mCRPC, Grade 3–4 adverse events were significantly lower with Lu-177 PSMA (33% vs 53%) — confirming a substantially better tolerability profile than the chemotherapy alternative.
Infrastructure Requirements in India
Accessing Lu-177 PSMA therapy requires infrastructure that not every hospital currently has. Before referring a patient, confirm the treating centre has:
- AERB licensing for radiopharmaceutical storage, handling, and administration
- PSMA PET/CT scanner — for mandatory pre-treatment patient selection (Gallium-68 or Fluorine-18 PSMA tracer)
- SPECT/CT capability — for post-dose biodistribution imaging and dosimetry
- Trained nuclear medicine team — physicians, radiopharmacists, radiation safety officer
- Lead-shielded preparation and administration areas — for radioactive compound handling
- Authorised pharmaceutical supply — genuine Lu-177 PSMA from licensed distributors with proper cold chain and radiation-safe packaging
India is actively expanding its nuclear medicine infrastructure — with major centres in Delhi, Mumbai, Chennai, Bangalore, Kolkata, and Hyderabad increasingly equipped to offer radioligand therapy.
A.K. Pharma — Prostate Cancer Medicine Distributor in Delhi
A.K. Pharma is a licensed Pharmaceutical distributor in Delhi supplying specialty prostate cancer medicines to hospitals, oncology centres, nuclear medicine facilities, and pharmacies across India.
Our complete prostate cancer portfolio includes every medicine that precedes, accompanies, and follows Lu-177 PSMA therapy in the treatment pathway:
- Glenza (Enzalutamide) — ARPI — required before Lu-177 PSMA eligibility
- Abirapro (Abiraterone Acetate) — CYP17 inhibitor — first-line mCRPC and mHSPC
- Zoladex (Goserelin) — GnRH agonist — mandatory ongoing ADT during Lu-177 PSMA treatment
- Firmagon (Degarelix) — GnRH antagonist — no testosterone flare, no antiandrogen cover needed
- Andocur (Cyproterone Acetate) — for antiandrogen flare cover and hot flush management
Browse our complete cancer medicines or contact us directly.
📍 E-2/257A, 2nd Floor, Shastri Nagar, New Delhi 110052
📞 011 4172 6999
📱 WhatsApp: +91 9810034827
Key Takeaways for Indian Oncologists and Patients
- February 2026 ASCO GU data confirms real-world median PFS of 13.5 months with Lu-177 PSMA therapy in taxane-naive mCRPC — consistent with PSMAfore trial
- PSMAddition trial supports potential approval for hormone-sensitive prostate cancer later in 2026 — significantly expanding eligible patient numbers
- PSMA PET/CT is mandatory before treatment — arrange imaging early in the treatment pathway
- Ongoing ADT is non-negotiable — Zoladex or Firmagon must be continued throughout all treatment cycles
- Nuclear medicine facility required — treatment cannot be given in standard oncology settings
- India is expanding RLT access — major centres across Delhi, Mumbai, Chennai, and Bangalore are building nuclear medicine capability
Frequently Asked Questions
Q. Is Lu-177 PSMA therapy the same as chemotherapy?
No — it is mechanistically completely different. Chemotherapy kills rapidly dividing cells non-specifically. Lu-177 PSMA therapy delivers targeted radiation selectively to PSMA-expressing prostate cancer cells throughout the body while largely sparing normal tissue. It is far better tolerated than taxane chemotherapy in most patients.
Q. Can Lu-177 PSMA therapy be used if the cancer has spread to bones?
Yes — bone metastases express PSMA and are targeted by Lu-177 PSMA therapy. The treatment is effective across bone metastases, lymph node deposits, and soft tissue disease simultaneously. Adequate bone marrow reserve must be confirmed before each cycle.
Q. Is Enzalutamide (Glenza) or Abiraterone (Abirapro) required before Lu-177 PSMA therapy?
Yes — prior ARPI therapy is required under current eligibility criteria. Patients must have received Enzalutamide or Abiraterone before being eligible for Lu-177 PSMA therapy under the PSMAfore-based approval.
Q. What medicines are needed alongside Lu-177 PSMA therapy?
Ongoing androgen deprivation therapy — Zoladex (Goserelin) or Firmagon (Degarelix) — must be continued throughout all 6 treatment cycles. Testosterone suppression must be maintained throughout.
Q. How can A.K. Pharma help with prostate cancer medicine supply?
A.K. Pharma supplies the complete prostate cancer medicine portfolio — Glenza, Abirapro, Zoladex, Firmagon, and Andocur — to hospitals and oncology centres across Delhi and India. Contact us at 011 4172 6999 or WhatsApp +91 9810034827.