In December 2019 the FDA approved a new cancer medicine that most oncologists had never heard of. By 2024 it had become the best-selling antibody-drug conjugate in the world — generating USD 3.75 billion in annual sales. By early 2026 it had received Priority Review for yet another new indication and was being described by leading oncologists as the most transformative cancer medicine developed in the past decade.
The medicine is Enhertu (Trastuzumab Deruxtecan). And the technology behind it — antibody-drug conjugates — is reshaping cancer treatment faster than any other development in oncology today.
The global ADC market, valued at USD 15.29 billion in 2025, is projected to reach USD 18.69 billion in 2026 — and USD 28.41 billion by 2035. For Indian oncologists, haematologists, and their patients, understanding what ADCs are, why Enhertu is leading this revolution, and what it means for cancer care in India has never been more important.
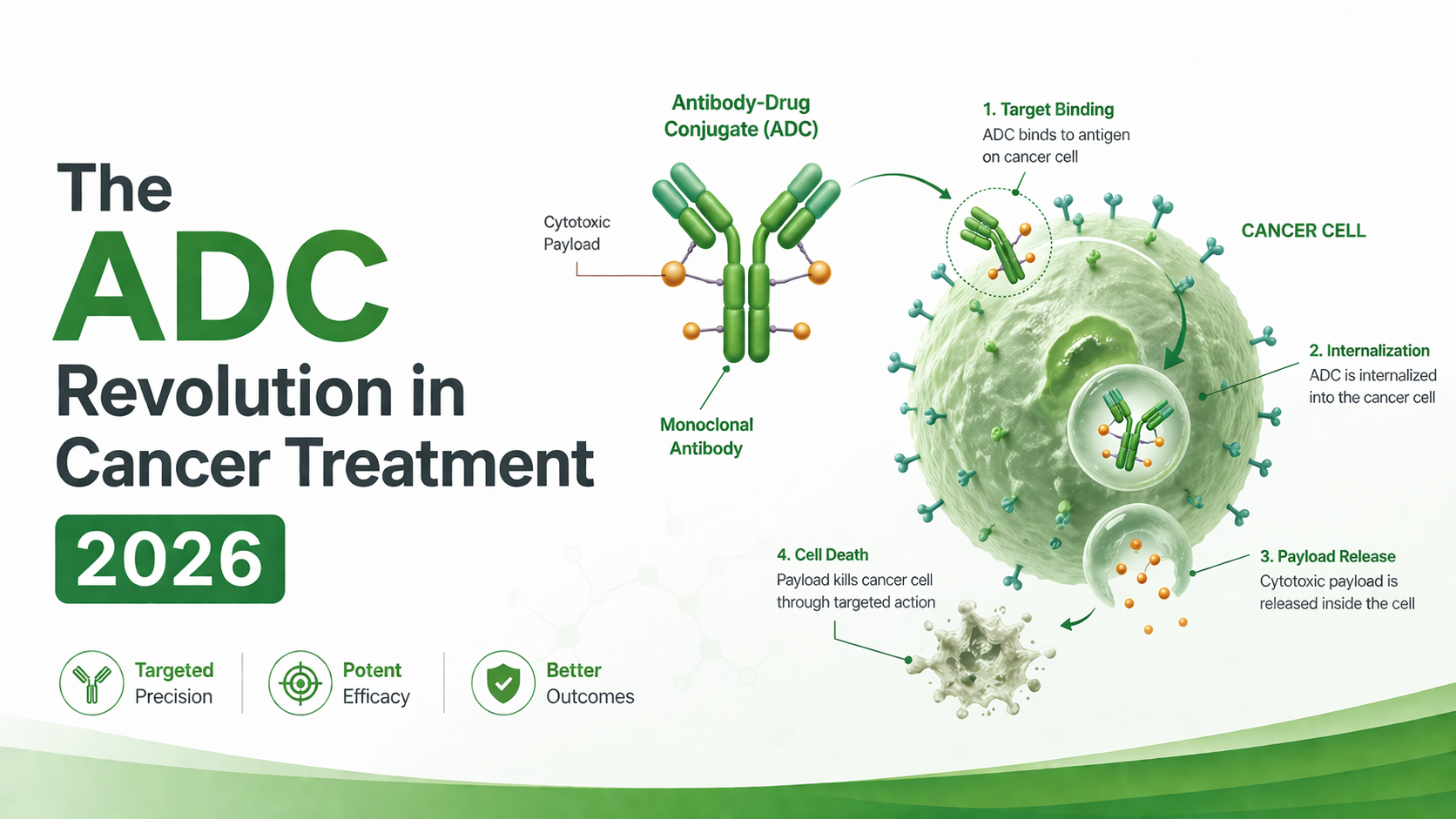
What is an Antibody-Drug Conjugate?
An antibody-drug conjugate (ADC) is a precisely engineered cancer medicine consisting of three components working together:
The first is a monoclonal antibody — a protein engineered to recognise and bind to a specific target on cancer cell surfaces with high precision. The second is a cytotoxic payload — a highly potent cancer-killing drug that would be too toxic to administer directly into the bloodstream. The third is a chemical linker — connecting the antibody to the payload in a way that keeps the payload stable in circulation but releases it selectively inside cancer cells.
The result is what oncologists describe as a “smart bomb” for cancer — the antibody acts as a homing device, finding cancer cells and delivering the cytotoxic payload directly inside them, while leaving healthy cells largely unaffected.
This concept has been under development since the 1980s — but earlier ADCs were limited by instability, premature payload release, low potency, and manufacturing challenges. The field lay largely dormant until a new generation of ADC technology emerged in the 2010s — with Enhertu at the forefront.
Why Enhertu Changed Everything
When Enhertu’s developer Daiichi Sankyo designed Trastuzumab Deruxtecan, they built it differently from earlier ADCs in three ways that proved decisive:
Higher payload density. Earlier HER2-targeted ADCs carried approximately 3-4 payload molecules per antibody. Enhertu carries approximately 8 — delivering roughly twice the cytotoxic payload per antibody molecule to the tumour.
More potent payload. Enhertu’s DXd payload is a topoisomerase I inhibitor approximately 10 times more potent than the active metabolite of irinotecan — a standard chemotherapy agent. This superior potency means more cancer cell death per payload molecule released.
Cleavable linker with bystander effect. After Enhertu is internalised into a cancer cell and the linker is cleaved by lysosomal enzymes, the released DXd payload is membrane-permeable — it diffuses out of the targeted cancer cell and kills neighbouring tumour cells regardless of whether they express HER2. This bystander killing effect is absent in earlier ADCs and is a key reason for Enhertu’s activity in tumours with heterogeneous HER2 expression.
These three design advances translated into clinical results that stunned the oncology community.
The DESTINY-Breast03 Result
In the DESTINY-Breast03 trial — a head-to-head comparison of Enhertu vs T-DM1 (Kadcyla), the previous standard second-line HER2-positive metastatic breast cancer therapy — Enhertu achieved a median progression-free survival of 28.8 months. T-DM1 achieved 6.8 months.
A 22-month difference in median PFS in a head-to-head oncology trial is extraordinary. The objective response rate with Enhertu was 79.7% — nearly double T-DM1’s 34.2%. The result rendered T-DM1 clinically obsolete in second-line HER2-positive metastatic breast cancer within months of publication.
To put the 28.8-month PFS in context — the previous generation of HER2-targeted therapy, Lapatinib + Capecitabine, achieved a median PFS of approximately 8 months in second-line HER2-positive disease. Enhertu improved on that by more than 20 months.
HER2-Low — Enhertu Created an Entirely New Cancer Category
DESTINY-Breast04 achieved something arguably more important than DESTINY-Breast03 — it created an entirely new treatable patient population.
Before DESTINY-Breast04, breast cancers were classified as either HER2-positive (approximately 20% of metastatic breast cancer) or HER2-negative — and HER2-negative patients were ineligible for any HER2-targeted therapy. DESTINY-Breast04 demonstrated that Enhertu significantly improved both progression-free survival and overall survival in patients with HER2-low (IHC 1+ or IHC 2+/ISH–) metastatic breast cancer. Median OS improved from 17.5 months with chemotherapy to 23.9 months with Enhertu.
Approximately 45-55% of all metastatic breast cancer patients are HER2-low. This single trial expanded the population eligible for HER2-targeted therapy from 20% to approximately 60% of all metastatic breast cancer patients. Oncology guidelines worldwide were updated. Pathologists began re-examining HER2-negative tumours for HER2-low status.
Enhertu Moving Into Early Breast Cancer — Priority Review in 2026
AstraZeneca and Daiichi Sankyo’s supplemental Biologics License Application for Enhertu has been accepted and granted Priority Review in the US for patients with HER2-positive breast cancer who have residual invasive disease after neoadjuvant HER2-targeted treatment — with the FDA action date anticipated during Q3 2026.
This post-neoadjuvant setting represents a high-risk population with significant unmet need. If approved, Enhertu would move from metastatic treatment into early-stage treatment — potentially improving cure rates for patients who currently face high recurrence risk. The DESTINY-Breast05 data earned Enhertu Breakthrough Therapy Designation in this setting — the FDA’s signal that a medicine is likely to offer a substantial improvement over existing options.
Beyond Breast Cancer — HER2-Mutant Lung Cancer and Gastric Cancer
In HER2-positive gastric and gastroesophageal junction adenocarcinoma, DESTINY-Gastric01 demonstrated Enhertu significantly improved overall survival — median OS 12.5 months vs 8.4 months with chemotherapy.
In HER2-mutant NSCLC — approximately 2-4% of all NSCLC patients who had no previously approved targeted option — Enhertu achieved a confirmed ORR of 49% in DESTINY-Lung02, with median PFS 9.9 months. Enhertu is the first and only approved targeted therapy for this patient population.
What This Means for Indian Oncology
AstraZeneca India Pharma Ltd. formally introduced Trastuzumab Deruxtecan in India in January 2024 for adults with metastatic or unresectable HER2-positive breast cancer who had already received an anti-HER2 regimen.
India has approximately 178,000 new breast cancer diagnoses annually — with approximately 20-25% being HER2-positive and a further 45-55% being HER2-low. The combined eligible population for Enhertu in India is substantial.
Hospitals and oncology centres requiring genuine Enhertu (Trastuzumab Deruxtecan) can contact A.K. Pharma — a trusted medicine distributor in Delhi supplying genuine imported oncology medicines to hospitals and pharmacies across India.
The Broader ADC Pipeline
At World ADC London 2026, experts highlighted how advances in payload design, targeting strategies, and AI-driven discovery are changing ADC development. Over 530 global ADC therapy programmes are under development across 140+ developers, with nearly 560 clinical trials including 190 evaluating ADC combinations.
Also available at A.K. Pharma: Trodelvy (Sacituzumab Govitecan) — a TROP-2 targeted ADC for triple-negative and HR-positive HER2-negative breast cancer.
Key Clinical Facts — Enhertu in 2026
- Standard second-line therapy for HER2-positive metastatic breast cancer
- Approved for HER2-low metastatic breast cancer — IHC 1+ or IHC 2+/ISH–
- Approved for HER2-positive gastric/GEJ adenocarcinoma after prior Trastuzumab
- First approved targeted therapy for HER2-mutant NSCLC
- Priority Review granted for post-neoadjuvant HER2-positive early breast cancer — FDA action date Q3 2026
- HER2 testing mandatory — IHC; ISH for IHC 2+; NGS for NSCLC
- ILD occurs in approximately 10-15% of patients — mandatory monitoring throughout
- Dilute only in 5% Dextrose — never in NaCl
This article is for informational purposes only. All prescribing decisions should be made by qualified oncologists based on individual patient assessment.
References
- DESTINY-Breast03 — Cortés J et al. NEJM (2022)
- DESTINY-Breast04 — Modi S et al. NEJM (2022)
- DESTINY-Lung02 — Li BT et al. JCO (2023)
- DESTINY-Gastric01 — Shitara K et al. NEJM (2021)
- Enhertu Priority Review — AstraZeneca (March 2026)
- ADC Market 2026 — Vision Lifesciences (February 2026)
- World ADC London 2026 — Drug Target Review (April 2026)